-
Notifications
You must be signed in to change notification settings - Fork 6
Home
Download BMDExpress 2 Software
New Features in BMDExpress 2 Build 3
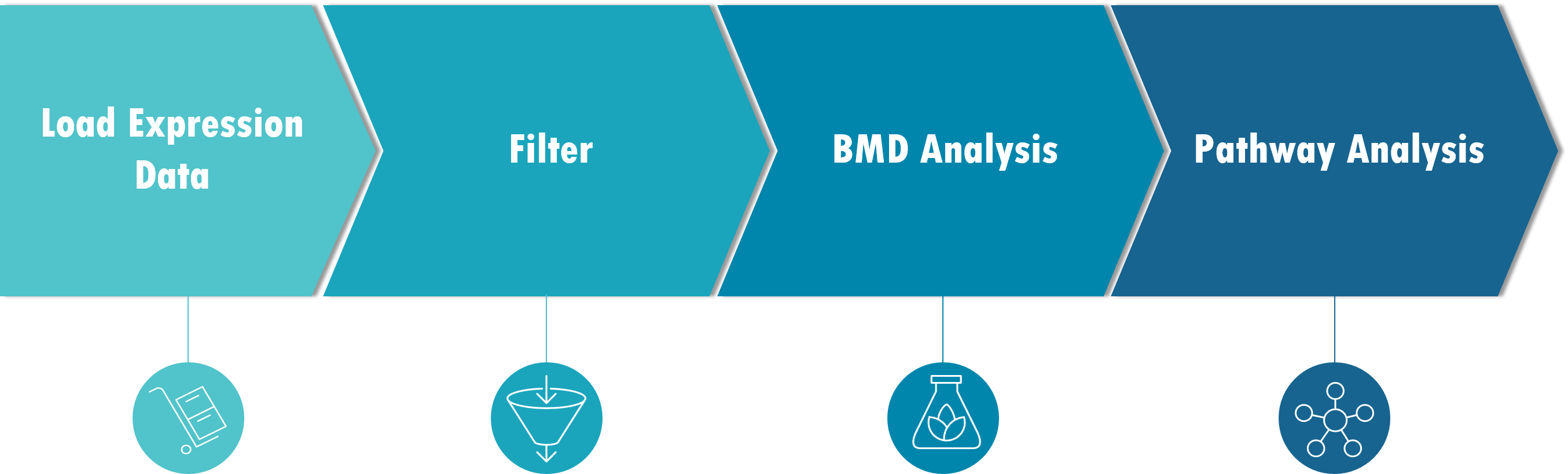
BMDExpress 2 is a desktop application for Windows, Mac or Linux (See IMPORTANT warning) that enables analysis of dose-response data produced in differential gene expression experiments. It provides stepwise workflows that combine benchmark dose (BMD) calculations with functional classification analysis based on the combined probesets for individual genes, Gene Ontology (GO), Signaling Pathways (Reactome, BioPlanet), or custom categories provided by the user. The end results are estimates of doses at which cellular processes are altered, based on an increase or decrease in response in expression levels compared to untreated controls. These estimates depend on fitting curves through the user's dose-response data. BMDExpress 2 utilizes both parametric and non-parametric curve (new in build 3) fitting methods. In build 3 IVIVE has been implemented which allows the user to convert benchmark concentration values from in vitro studies into estimated external doses (i.e., oral equivalent doses).
Parametric: US EPA BMDS software
Non-parametric: Sciome GCurveP
Examples:
Example BMDExpress 2 expression data files
Note: Compressed zip file that contains 3 expression data files. User will need to unzip before using.
Example BMDExpress 2 project file (.bm2)
Note: Compressed zip file that contains the .bm2 file used in the tutorial videos. User will need to unzip the file before loading into BMDExpress 2.
Before working with data, verify that necessary gene annotations are present, and up to date. Annotations for the various genomic platforms are stored and maintained on the BMD Express 2 GitHub site. Before importing data, annotations must be present locally.
Gene expression data is first imported into BMDExpress. The data must be correctly formatted. Excel files are not currently supported, but tab-delimited .txt files are supported. Data sets can be prefiltered outside of BMDExpress (i.e., features can be removed through statistical filtering), and a subset of the data can be loaded and modeled. Alternatively the data set can be loaded in its entirety and filtered within BMDExpress (see below).
Gene expression dose-response data is then (optionally) processed using one of several choices of statistical model, together with a fold change filter to identify probes/probe sets that demonstrate dose-response behavior in accordance with user-specified thresholds. Filtering the probe sets for such a threshold in dose-response behavior is not required, but will reduce noise in the data and the computation time required in the subsequent steps in the analysis.
Dose response data is then fit to one of the following models:
- parametric
- Power
- Linear
- Polynomial 2-4
- Hill
- Exponential 2-5
- non-parametric
- gCurveP
In the case of parametric modeling, the model that best describes the data without having too much complexity is selected for subsequent procedures. The user can apply two approaches for paremetric model selection including 1) a nested likelihood ratio test for the linear and polynomial models followed by an Akaike information criterion (AIC) that compares the best nested model to the exponential model, Hill model and the power model; or 2) a completely AIC-based selection process to compare all models.
Non-parametric gCurveP produces a single output, based on the choice of input parameters.
After modeling is complete (best model in the case of parametric), probe/probeset identifiers are mapped onto unique genes based on NCBI Entrez Gene identifiers. Entrez Gene IDs are subsequently matched to corresponding Gene Ontology, Signaling Pathway (e.g., Reactome), or user defined categories. Summary values representing the central tendencies and associated variability of the BMD, benchmark dose lower confidence limits (BMDL) and benchmark dose upper confidence limits (BMDU) for all the genes in each category are then computed.
Batchwise processing of multiple data sets is available at every step of the workflow. This is accomplished by standard multi-select keyboard/mouse click combinations specific to the host operating system.
Results can then be exported for further analysis in other software packages.
Project files from BMDExpress 2 are saved in .bm2 format or alternatively in .json format (much larger than .bm2). However, .bmd project files from the original BMDExpress can be imported in to the software and transformed into the .bm2 format. Note: .bmd files loaded into BMDExpress retain all annotations and results contained in the original file, hence, it will appear that there is missing data in the results files and probe annotations may not match updated annotations that would be applied if the expression data was re-analyzed in BMDExpress 2.
BMDExpress 2 can analyze any continuous dose-response data. An example video on how to perform analysis on nongenomic data in BMDExpress 2 can be found here.
Finally, IVIVE is an adjunct functionality provided with BMDExpress 2 as a convience for additional post-processing of in vitro transcriptomic data.
A playlist of video tutorials created by Scott Auerbach is available. Videos will also be linked in each section for their relevant functions.
During the BMDS analysis, the EPA BMDS command line executables require gfortran libraries to be installed on your Mac. If these libraries are not installed on your Mac, then there will be a bunch of -9999 values in the results table that were returned by the EPA BMDS command line executables. The EPA BMDS executables were compiled under the assumption that the gfortran libraries are located in the folder: /usr/local/gfortran/lib
In case you have do not have the gfortran libraries installed, we offer a workaround solution outlined below. A video tutorial on how to install the gfortran libraries is also available.
- Open Terminal
- Create the directory that will contain the gfortran libs:
sudo mkdir -p /usr/local/gfortran/lib - Download the gfortran libraries: Click here to download MacOS gfortran libraries
- Inside the Terminal, change directory to Downloads:
cd Downloads - Unzip the downloaded file:
unzip mac_gfortran_deps.zip - Copy libgfortran.3.dylib to gfortran lib directory:
sudo cp libgfortran.3.dylib /usr/local/gfortran/lib/ - Copy libquadmath.0.dylib to gfortran lib directory:
sudo cp libquadmath.0.dylib /usr/local/gfortran/lib/
Disclaimer: It is your responsibility to perform these actions in compliance with the security protocols of your organization. As with any file downloaded from the internet, you are highly encouraged to perform a virus scan on these files after you download them to ensure they have not been tampered with during the data transfer process.