k_low and T1, T3 corrected for KLI_GLB_2016, NOx2018; PdepArrhenius info added for Nitrogen_GLB_JMZ#612
Conversation
There was a problem hiding this comment.
@agi-jaguar Thanks for making the correction. I checked the predicted ks from these new numbers and they look pretty close to the ks from Jasper et al. (original source https://pubs.acs.org/doi/full/10.1021/jp067585p).
One suggestion before merging this in. Can you add a comment in LongDesc (add it after the shortDesc about this change...). In the comment, I think you should mention:
- These entries have different numbers than the entries in the original mechanism files from the KG2016 and NOx2018 paper. Include the original numbers in the KG2016, NOx2018.
- The reason about the change. (E.g., these numbers are adapted from Jasper et al., but the authors missed multiplying Na.)
In the PR, can you also include a plot showing how this changes the prediction of ks, and probably compared with the ks from Jasper et al.? This can be a good reference for future developers about this data change.
|
I also made changed to Nitrogen_Glaborg_Jimenez as below The reference of this reaction (Davis et al. , J. Phys. Chem. A 1999, 103, 5889-589910.1021/jp982762a) reported PdepArrhenius reaction information. However, RMG database (Nitrogen_Glaborg_Jimenez_ only contained the rate coefficient at 1 atm, so reported all of them (0.1, 1.0, 10, 100 atm) |
|
@agi-jaguar Sorry that I changed my review. Can you
I will approve your PR and merge it afterward. |
 xiaoruiDong
left a comment
xiaoruiDong
left a comment
There was a problem hiding this comment.
See my last comments
 xiaoruiDong
left a comment
xiaoruiDong
left a comment
There was a problem hiding this comment.
Thank you for finding another issue. Plz see my comments
| pressures = ([0.01, 1, 10, 20, 50, 100], 'atm'), | ||
| arrhenius = [ | ||
| Arrhenius(A=(6.8e+24, 'cm^3/(mol*s)'), n=-3, Ea=(0, 'cal/mol'), T0=(1, 'K')), | ||
| Arrhenius(A=(8.3476e+20, 'cm^3/(mol*s)'), n=-4, Ea=(0, 'cal/mol'), T0=(1, 'K')), |
There was a problem hiding this comment.
Can you add a description of the change you made (basically, *P/RT) and a reference to issue #613 in the new commit you added, so the future developer has a better understanding of the change?
a687655 to
788b864
Compare
| shortDesc = u"""The chemkin file reaction is CH3OH <=> CH2(S) + H2O""", | ||
| longDesc = u"""These entries have different numbers than the entries in the original mechanism files | ||
| from the Jasper et al., 2007. A factors for low pressure limit should be multiplied by avogadro's | ||
| number since the number in the original paper has the unit of molecule rather than mol )""" |
There was a problem hiding this comment.
I noticed a parenthesis was forgotten to remove at the end. Plus, I just realized that T1 and T3 issue was not mentioned. Maybe consider the following paragraph?
longDesc = u"""This entry has different numbers than the entry in the mechanism file (Hashemi et al.).It uses the kinetic parameters from the original source (Jasper et al. J. Phys. Chem. A, 2007, 111, 19, 3932–3950). Jasper et al. reported kinetic unit in molecule, cm^3, and s, but Hashemi et al. didn't multiply the A factor of the low-pressure limit Arrhenius by Avogadro's number when compiling the mechanism (with the unit in mol, cm^3, and s). Besides, Hashemi et al. mistakenly reversed the order of the T1 and T3 parameters."""
| ), | ||
| longDesc=u"""low pressure limit k_0 was taken from Fernandes et al., 2006. However, it has to | ||
| be noted that [M] should be multiplied to 7E-31300^3T^-3 [cm6/#2/s]. Thus, | ||
| k_0~ 7E-31300^3T^-3 [cm6/#2/s] * P/RT [#/cm3] = 7E-31300^31013.25/8.314E6*(6.02E23)^2 [cm6/#/s] = 8.348E+20*T^-4""" |
There was a problem hiding this comment.
I suggest the following. There are minor mistakes in the long description, and the formula is hard to follow.
longDesc = u"""This entry has different rate constants at P=0.01 atm than the entry in the mechanism file (Glarborg et al.). In Glarborg et al., k at 0.01 atm was taken from the low-pressure limit k0 of the Troe kinetics in Fernandes et al. (Fernandes et al., JPCA, 110, 13, 4442-4449). However, according to the definition by Fernandes et al., k0 (7E-31*(T/300)^-3 [cm6/#2/s]) has to be multiplied by [M] (P/RT) to get the rate constants at a specific pressure. Thus, k(P=0.01 atm) ~ 7E-31*(T/300)^-3 [cm6/#2/s] * P/RT [mol/cm3] = 7E-31*(300^3)(6.02E23)^2(1013.25/8.314E6)(T^-4) [cm3/mol/s] = 8.348E+20T^-4"""
| shortDesc = u"""The chemkin file reaction is CH3OH <=> CH2(S) + H2O""", | ||
| longDesc = u"""These entries have different numbers than the entries in the original mechanism files | ||
| from the Jasper et al., 2007. A factors for low pressure limit should be multiplied by avogadro's | ||
| number since the number in the original paper has the unit of molecule rather than mol )""" |
There was a problem hiding this comment.
longDesc = u"""This entry has different numbers than the entry in the mechanism file (Hashemi et al.). It uses the kinetic parameters from the original source (Jasper et al. J. Phys. Chem. A, 2007, 111, 19, 3932–3950). Jasper et al. reported kinetic unit in molecule, cm^3, and s, but Hashemi et al. didn't multiply the A factor of the low-pressure limit Arrhenius by Avogadro's number when compiling the mechanism (with the unit in mol, cm^3, and s). Besides, Hashemi et al. mistakenly reversed the order of the T1 and T3 parameters."""
| Ea = (37600, 'cal/mol'), | ||
| T0 = (1, 'K'), | ||
| ), | ||
| ], |
There was a problem hiding this comment.
Sorry Jeehyun @agi-jaguar , do you have some time to also add a description here? Thanks.
LongDesc="""This entry has different kinetics than the entry in the mechanism file (Lopez et al.). The original reference of this reaction (Davis et al. , J. Phys. Chem. A 1999, 103, 5889-5899) reported Pdep kinetics. However, the kinetics in the mechanism file of Lopez et al. only contained the rate coefficient at 1 atm and used it as the high-pressure limit rate coefficient. This entry instead reported the Pdep kinetics from the original source."""
There was a problem hiding this comment.
No worries Jerry! I added the description!
 xiaoruiDong
left a comment
xiaoruiDong
left a comment
There was a problem hiding this comment.
Hi @agi-jaguar, Thank you so much for helping correct these numbers. It is really helpful! I left a few comments about correcting the description. I hope you understand that I want to make sure all the changes are trackable, understandable, and reproducible by future developers, and your effort is highly appreciated.
update C2H2+CH3=CH2CHCH2 with pdep kinetics
description added
longdesc_error_fix
|
@hwpang I have already checked the correction proposed by Jeehyun @agi-jaguar. However, Jeehyun is very busy and doesn't have time to update some of the descriptions I suggested. Therefore, I pushed a new commit to add more details to describe the correction of each modified entry in the corresponding "long description". Please let me know if you think these comments read okay and are understandable by other (potentially future) developers. Thanks! |
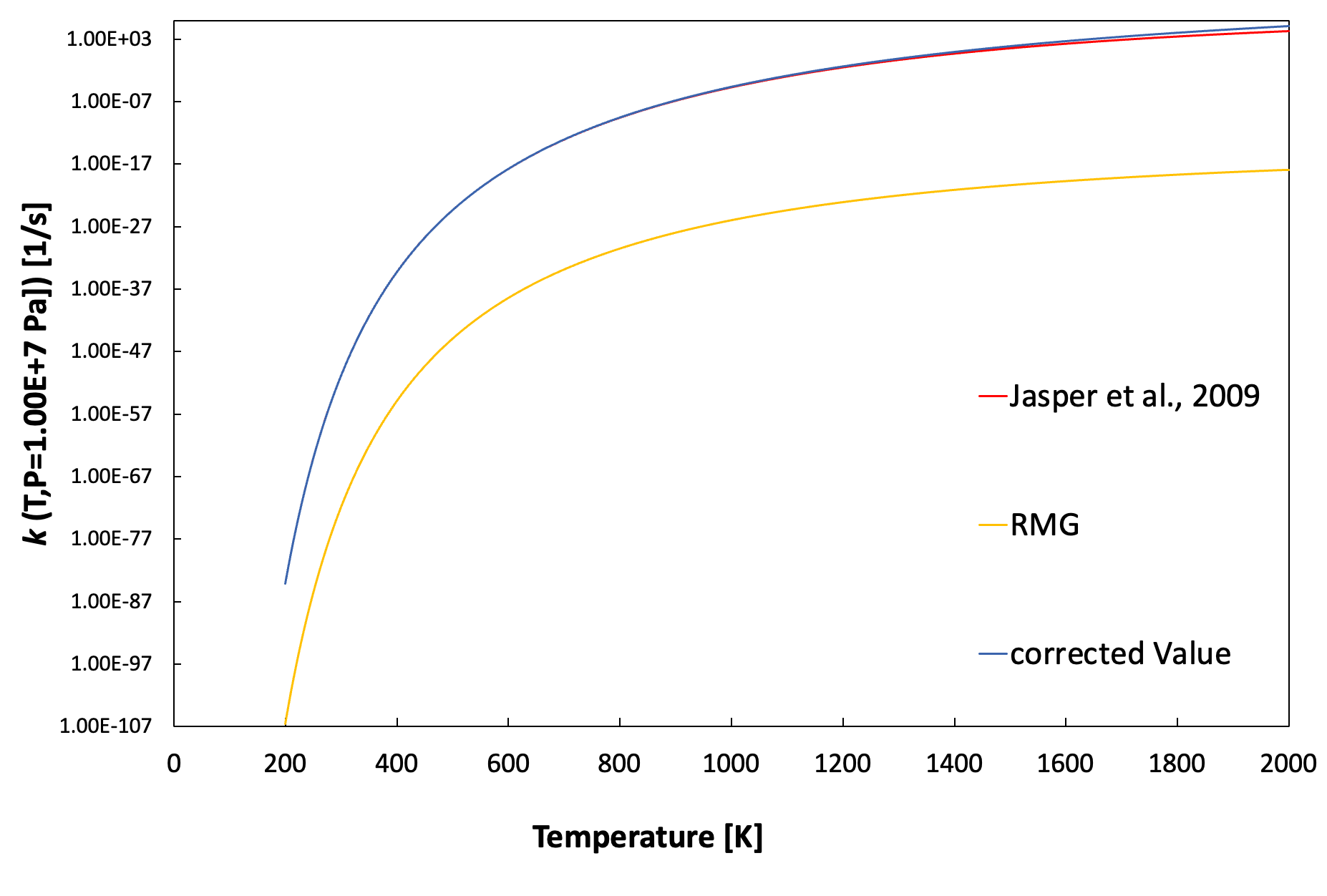
Klippenstein_Glaborg_2016 and NOx_2018 CH3OH -> CH2(S) + H2O's low pressure k has been corrected. Also, T1 and T3 for Troe format have been corrected